Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 to obtain the high U solution
to obtain the high U solution; *; Sakurai, Koji*; ; Nomura, Kazunori; *
JNC TN8400 2000-032, 98 Pages, 2000/12
Concerning the preparation of high U solution for the crystallization process and the application of UO powder dissolution to that, the effects of final U concentration, dissolution temperature, nitric acid concentration and powder size on the dissolution of UO
powder dissolution to that, the effects of final U concentration, dissolution temperature, nitric acid concentration and powder size on the dissolution of UO powder in the nitric acid where the final U concentration was
powder in the nitric acid where the final U concentration was  800g/L were investigated. The experimental results showed that the solubility of UO
800g/L were investigated. The experimental results showed that the solubility of UO decreased with the increase of final UO
decreased with the increase of final UO concentration and powder size, and with the decrease of dissolution temperature and nitric acid concentration. It was also confirmed that in the condition where the final U concentration was sufficiently lower than the solubility of U, UO
concentration and powder size, and with the decrease of dissolution temperature and nitric acid concentration. It was also confirmed that in the condition where the final U concentration was sufficiently lower than the solubility of U, UO dissolution behavior in the high U solution could be estimated with the equation based on the fragmentation model which we had already reported. Based on these experimental results, the dissolution behavior of irradiated MOX fuel in high U solution was estimated and the possibility of supplying high U solution to the crystallization process was discussed. In the preparation of high U solution for the crystallization process, it was estimated that the present dissolution process (dissolution for fuel pieces of about 3cm long) needed a lot of time to obtain a high dissolution yield, but it was shorted drastically by the pulverization of fuel pieces. The burst of off-gas at the early in the dissolution of fuel powder seems to be avoidable with setting the appropriate dissolution condition, and it is important to optimize the dissolution condition with considering the capacity of off-gas treatment process.
dissolution behavior in the high U solution could be estimated with the equation based on the fragmentation model which we had already reported. Based on these experimental results, the dissolution behavior of irradiated MOX fuel in high U solution was estimated and the possibility of supplying high U solution to the crystallization process was discussed. In the preparation of high U solution for the crystallization process, it was estimated that the present dissolution process (dissolution for fuel pieces of about 3cm long) needed a lot of time to obtain a high dissolution yield, but it was shorted drastically by the pulverization of fuel pieces. The burst of off-gas at the early in the dissolution of fuel powder seems to be avoidable with setting the appropriate dissolution condition, and it is important to optimize the dissolution condition with considering the capacity of off-gas treatment process.
Takachi, Kazuhiko; Suzuki, Hideaki*
JNC TN8400 99-041, 76 Pages, 1999/11
The buffer material is expected to maintain its low water permeability, self-sealing properties, radionuclides adsorption and retardation properties, thermal conductivity, chemical buffering properties, overpack supporting properties, stress buffering properties, etc. over a long period of time. Natural clay is mentioned as a material that can relatively satisfy above. Among the kinds of natural clay, bentonite when compacted is superior because (1)it has exceptionally low water permeability and properties to control the movement of water in buffer, (2)it fills void spaces in the buffer and fractures in the host rock as it swells upon water uptake, (3)it has the ability to exchange cations and to adsorb cationic radioelements. In order to confirm these functions for the purpose of safety assessment, it is necessary to evaluate buffer properties through laboratory tests and engineering-scale tests, and to make assessments based on the ranges in the data obtained. This report describes the procedures, test conditions, results and examinations on the buffer material of unconfined compression tests, one-dimensional consolidation tests, consolidated-undrained triaxial compression tests and consolidated-undrained triaxial creep tests that aim at getting hold of static mechanical properties. We can get hold of the relationship between the dry density and tensile stress etc. by Brazillian tests, between the dry density and unconfined compressive strength etc. by unconfined compression tests, between the consolidation stress and void ratio etc. by one-dimensional consolidation tests, the stress pass of each effective confining pressure etc. by consolidated-undrained triaxial compression tests and the axial strain rate with time of each axial stress etc. by consolidated-undrained triaxial creep tests.
Sato, Haruo
JNC TN8400 99-064, 22 Pages, 1999/10
Four kinds of diffusion experiments; (1)through-diffusion(T-D) experiments for compaction direction dependency, (2)in-diffusion(I-D) experiments for composition dependency of silica sand in bentonite, (3)I-D experiments for initial bentonite gain size dependency, and (4)I-D experiments for the restoration property of an artificial single fracture in compacted bentonite, were carried out using tritiated water which is a non-sorbing nuclide to evaluate the effect of pore structural factors for eompacted bentonite on diffudion. For(1), effective diffusivities (De) in Na-bentonites, Kunigel-V1 and Kunipia-F were measured for 1.0 and 1.5 Mg m
m . For(2), apparent diffusivities (Da) in Kunigel-V1 were measured for 0.8, 1.4 and 1.8 Mg
. For(2), apparent diffusivities (Da) in Kunigel-V1 were measured for 0.8, 1.4 and 1.8 Mg m
m with silica sand of 30 and 50 wt%. For(3), Da values for 0.8, 1.4 and 1.8 Mg
with silica sand of 30 and 50 wt%. For(3), Da values for 0.8, 1.4 and 1.8 Mg m
m were measured for a granulated Na-bentonite, OT-9607 which grain-size distribution is in a rang between 0.1 and 5 mm. For (4), Da values in Kunigel-V1 which a single fracture was artificially reproduced and was immersed in distilled water for 7 or 28 days for the restoration of the fracture, were measured for 1.8 Mg
were measured for a granulated Na-bentonite, OT-9607 which grain-size distribution is in a rang between 0.1 and 5 mm. For (4), Da values in Kunigel-V1 which a single fracture was artificially reproduced and was immersed in distilled water for 7 or 28 days for the restoration of the fracture, were measured for 1.8 Mg m
m . Although De values in Kunigel-V1 were approximately the same for both compacted directions over the density, De values for perpendicular direction to compacted direction were higher than those for the same direction as compacted direction in Kunipia-F. For composition dependency of silica sand in bentonite, no significant effect of the mixure of silica sand in bentonite on Da was found. For initial bentonite grain size dependency, Da values obtained for OT-960 were approximately the same as those for Kunigel-V1 and no effect of initial grain size of bentonite on diffusion was found. For the restoration property of a single fracture in compacted bentonite, no restoration period dependency on Da was found. Based on this, it may be said that diffusion of nuclides in compacted bentonite, ...
. Although De values in Kunigel-V1 were approximately the same for both compacted directions over the density, De values for perpendicular direction to compacted direction were higher than those for the same direction as compacted direction in Kunipia-F. For composition dependency of silica sand in bentonite, no significant effect of the mixure of silica sand in bentonite on Da was found. For initial bentonite grain size dependency, Da values obtained for OT-960 were approximately the same as those for Kunigel-V1 and no effect of initial grain size of bentonite on diffusion was found. For the restoration property of a single fracture in compacted bentonite, no restoration period dependency on Da was found. Based on this, it may be said that diffusion of nuclides in compacted bentonite, ...
 -
- cascades in the decay of
cascades in the decay of 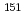 Nd
Nd; *; *
Journal of the Physical Society of Japan, 58(9), p.3143 - 3150, 1989/09
Times Cited Count:1 Percentile:23.68(Physics, Multidisciplinary)no abstracts in English
; ; ; ;
JAERI-M 7373, 25 Pages, 1977/11
no abstracts in English
; Tagawa, Hiroaki
Journal of Nuclear Materials, 71(1), p.65 - 72, 1977/01
Times Cited Count:31no abstracts in English